The Number of Electrons in the Valence Shell Determine What Types of Bonds an Atom Can Form
Affiliate vii. Chemical Bonding and Molecular Geometry
7.2 Covalent Bonding
Learning Objectives
By the terminate of this section, you volition be able to:
- Draw the germination of covalent bonds
- Define electronegativity and assess the polarity of covalent bonds
In ionic compounds, electrons are transferred between atoms of different elements to form ions. But this is not the merely mode that compounds can be formed. Atoms can also brand chemical bonds by sharing electrons as between each other. Such bonds are called covalent bonds. Covalent bonds are formed between two atoms when both have similar tendencies to concenter electrons to themselves (i.e., when both atoms take identical or fairly similar ionization energies and electron affinities). For case, ii hydrogen atoms bail covalently to form an Htwo molecule; each hydrogen atom in the H2 molecule has ii electrons stabilizing information technology, giving each atom the same number of valence electrons as the noble gas He.
Compounds that contain covalent bonds exhibit different concrete properties than ionic compounds. Because the allure betwixt molecules, which are electrically neutral, is weaker than that between electrically charged ions, covalent compounds generally take much lower melting and boiling points than ionic compounds. In fact, many covalent compounds are liquids or gases at room temperature, and, in their solid states, they are typically much softer than ionic solids. Furthermore, whereas ionic compounds are good conductors of electricity when dissolved in h2o, nigh covalent compounds are insoluble in water; since they are electrically neutral, they are poor conductors of electricity in whatever state.
Formation of Covalent Bonds
Nonmetal atoms oft form covalent bonds with other nonmetal atoms. For example, the hydrogen molecule, Hii, contains a covalent bond between its 2 hydrogen atoms. Effigy 1 illustrates why this bond is formed. Starting on the far right, we accept ii separate hydrogen atoms with a particular potential free energy, indicated past the ruddy line. Along the 10-axis is the altitude between the two atoms. As the two atoms approach each other (moving left along the x-centrality), their valence orbitals (ones) begin to overlap. The single electrons on each hydrogen atom and so collaborate with both atomic nuclei, occupying the space around both atoms. The strong attraction of each shared electron to both nuclei stabilizes the arrangement, and the potential energy decreases equally the bond distance decreases. If the atoms continue to approach each other, the positive charges in the two nuclei begin to repel each other, and the potential energy increases. The bond length is adamant by the distance at which the everyman potential energy is accomplished.
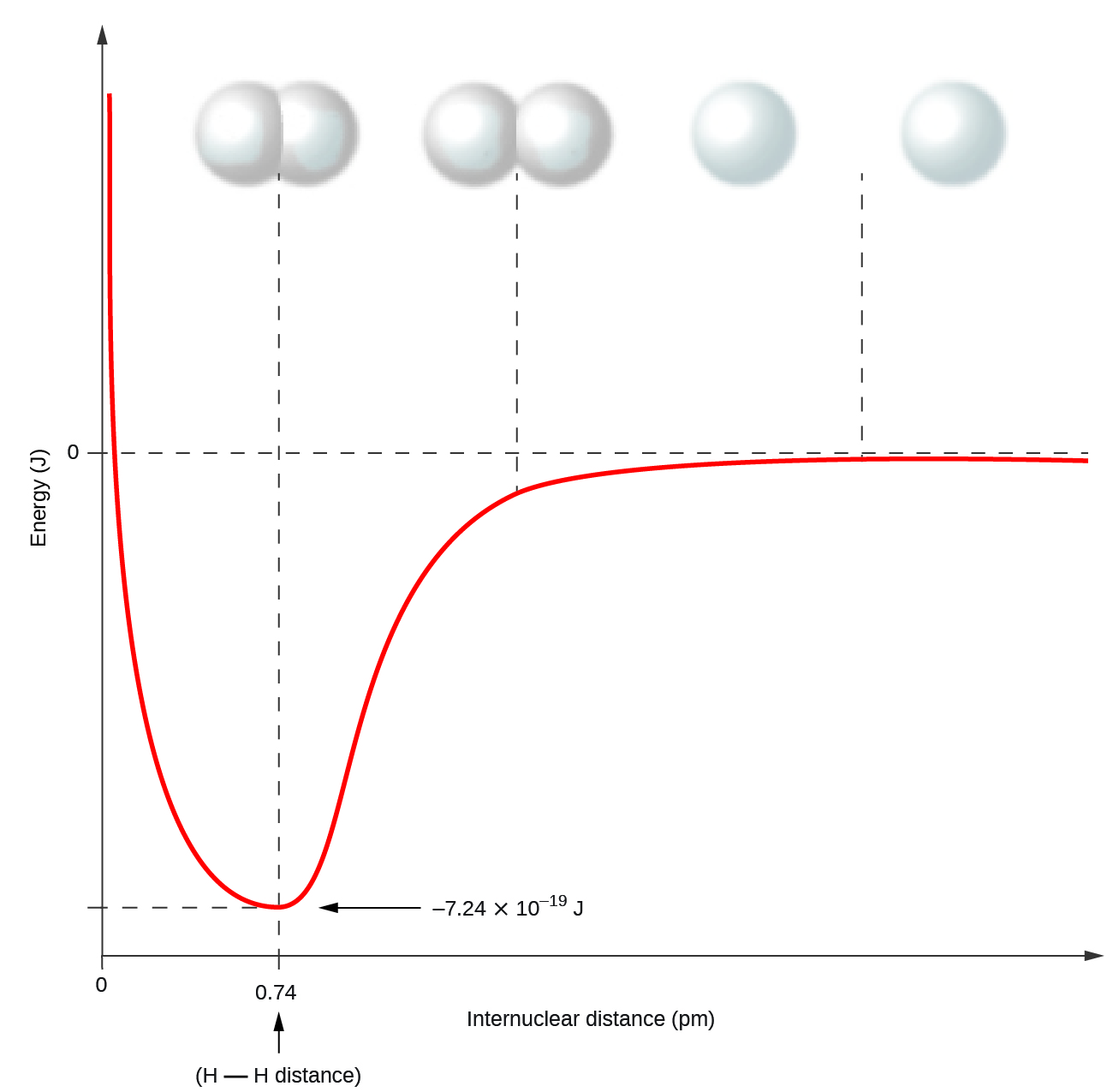
It is essential to think that energy must be added to break chemical bonds (an endothermic process), whereas forming chemical bonds releases free energy (an exothermic procedure). In the case of H2, the covalent bail is very strong; a large amount of energy, 436 kJ, must exist added to pause the bonds in i mole of hydrogen molecules and cause the atoms to divide:
[latex]\text{H}_2(g) \longrightarrow 2\text{H}(thousand) \;\;\;\;\; \Delta H = 436\;\text{kJ}[/latex]
Conversely, the same amount of energy is released when one mole of H2 molecules forms from two moles of H atoms:
[latex]2\text{H}(g) \longrightarrow \text{H}_2(g) \;\;\;\;\; \Delta H = -436 \;\text{kJ}[/latex]
Pure vs. Polar Covalent Bonds
If the atoms that form a covalent bond are identical, as in H2, Cltwo, and other diatomic molecules, then the electrons in the bond must be shared equally. We refer to this as a pure covalent bond. Electrons shared in pure covalent bonds take an equal probability of being nearly each nucleus.
In the case of Clii, each atom starts off with 7 valence electrons, and each Cl shares one electron with the other, forming one covalent bond:
[latex]\text{Cl} + \text{Cl} \longrightarrow \text{Cl}_2[/latex]
The total number of electrons around each individual atom consists of 6 nonbonding electrons and ii shared (i.e., bonding) electrons for viii full electrons, matching the number of valence electrons in the noble gas argon. Since the bonding atoms are identical, Clii also features a pure covalent bond.
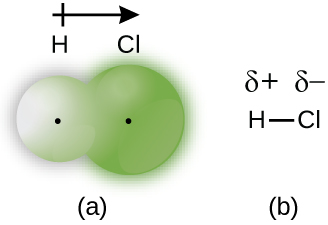
When the atoms linked past a covalent bond are dissimilar, the bonding electrons are shared, but no longer equally. Instead, the bonding electrons are more attracted to 1 cantlet than the other, giving rise to a shift of electron density toward that atom. This unequal distribution of electrons is known as a polar covalent bail, characterized past a partial positive charge on one atom and a partial negative charge on the other. The cantlet that attracts the electrons more strongly acquires the partial negative charge and vice versa. For example, the electrons in the H–Cl bond of a hydrogen chloride molecule spend more time near the chlorine cantlet than near the hydrogen cantlet. Thus, in an HCl molecule, the chlorine atom carries a partial negative charge and the hydrogen atom has a partial positive charge. Figure 2 shows the distribution of electrons in the H–Cl bond. Note that the shaded area effectually Cl is much larger than it is around H. Compare this to Effigy 1, which shows the even distribution of electrons in the H2 nonpolar bond.
We sometimes designate the positive and negative atoms in a polar covalent bail using a lowercase Greek letter "delta," δ, with a plus sign or minus sign to point whether the cantlet has a partial positive charge (δ+) or a fractional negative charge (δ–). This symbolism is shown for the H–Cl molecule in Figure 2.
Electronegativity
Whether a bond is nonpolar or polar covalent is determined by a belongings of the bonding atoms called electronegativity. Electronegativity is a measure of the trend of an atom to attract electrons (or electron density) towards itself. Information technology determines how the shared electrons are distributed between the two atoms in a bond. The more strongly an atom attracts the electrons in its bonds, the larger its electronegativity. Electrons in a polar covalent bond are shifted toward the more electronegative atom; thus, the more electronegative cantlet is the one with the partial negative charge. The greater the difference in electronegativity, the more polarized the electron distribution and the larger the partial charges of the atoms.
Figure iii shows the electronegativity values of the elements as proposed by ane of the most famous chemists of the twentieth century: Linus Pauling (Figure four). In general, electronegativity increases from left to correct across a period in the periodic table and decreases down a group. Thus, the nonmetals, which lie in the upper correct, tend to take the highest electronegativities, with fluorine the about electronegative element of all (EN = 4.0). Metals tend to be less electronegative elements, and the grouping ane metals accept the lowest electronegativities. Note that noble gases are excluded from this effigy considering these atoms ordinarily do non share electrons with others atoms since they take a full valence beat out. (While noble gas compounds such as XeOii exercise be, they can simply be formed under extreme conditions, and thus they practise not fit neatly into the general model of electronegativity.)

Electronegativity versus Electron Affinity
We must exist careful not to confuse electronegativity and electron affinity. The electron affinity of an element is a measurable physical quantity, namely, the energy released or absorbed when an isolated gas-phase atom acquires an electron, measured in kJ/mol. Electronegativity, on the other paw, describes how tightly an atom attracts electrons in a bond. It is a dimensionless quantity that is calculated, non measured. Pauling derived the first electronegativity values by comparing the amounts of free energy required to break different types of bonds. He chose an arbitrary relative scale ranging from 0 to 4.
Linus Pauling
Linus Pauling, shown in Figure four, is the but person to have received ii unshared (individual) Nobel Prizes: ane for chemical science in 1954 for his work on the nature of chemical bonds and one for peace in 1962 for his opposition to weapons of mass destruction. He developed many of the theories and concepts that are foundational to our current understanding of chemistry, including electronegativity and resonance structures.

Pauling also contributed to many other fields besides chemistry. His research on sickle jail cell anemia revealed the crusade of the disease—the presence of a genetically inherited aberrant protein in the claret—and paved the way for the field of molecular genetics. His work was also pivotal in curbing the testing of nuclear weapons; he proved that radioactive fallout from nuclear testing posed a public wellness take a chance.
Electronegativity and Bond Type
The absolute value of the deviation in electronegativity (ΔEN) of two bonded atoms provides a rough measure of the polarity to be expected in the bond and, thus, the bond type. When the divergence is very small or null, the bail is covalent and nonpolar. When it is large, the bond is polar covalent or ionic. The absolute values of the electronegativity differences betwixt the atoms in the bonds H–H, H–Cl, and Na–Cl are 0 (nonpolar), 0.ix (polar covalent), and two.1 (ionic), respectively. The degree to which electrons are shared between atoms varies from completely equal (pure covalent bonding) to not at all (ionic bonding). Effigy 5 shows the relationship betwixt electronegativity divergence and bond blazon.
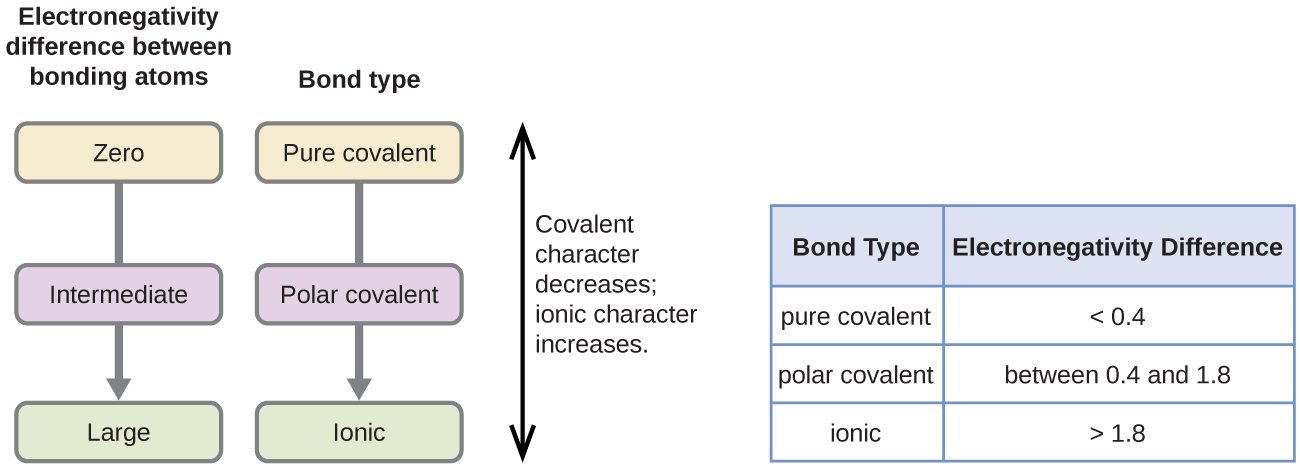
A rough approximation of the electronegativity differences associated with covalent, polar covalent, and ionic bonds is shown in Figure v. This table is just a general guide, however, with many exceptions. For example, the H and F atoms in HF take an electronegativity difference of 1.ix, and the N and H atoms in NH3 a difference of 0.9, yet both of these compounds form bonds that are considered polar covalent. Likewise, the Na and Cl atoms in NaCl have an electronegativity difference of ii.one, and the Mn and I atoms in MnItwo take a departure of 1.0, yet both of these substances form ionic compounds.
The all-time guide to the covalent or ionic character of a bond is to consider the types of atoms involved and their relative positions in the periodic table. Bonds between two nonmetals are generally covalent; bonding between a metal and a nonmetal is frequently ionic.
Some compounds incorporate both covalent and ionic bonds. The atoms in polyatomic ions, such equally OH–, NO3 −, and NH4 +, are held together by polar covalent bonds. However, these polyatomic ions form ionic compounds by combining with ions of opposite charge. For case, potassium nitrate, KNO3, contains the One thousand+ cation and the polyatomic NO3 − anion. Thus, bonding in potassium nitrate is ionic, resulting from the electrostatic attraction between the ions K+ and NO3 −, as well every bit covalent between the nitrogen and oxygen atoms in NO3 −.
Instance ane
Electronegativity and Bond Polarity
Bail polarities play an important function in determining the structure of proteins. Using the electronegativity values in Figure iii, arrange the following covalent bonds—all commonly plant in amino acids—in order of increasing polarity. Then designate the positive and negative atoms using the symbols δ+ and δ–:
C–H, C–N, C–O, Due north–H, O–H, Due south–H
Solution
The polarity of these bonds increases as the absolute value of the electronegativity difference increases. The atom with the δ– designation is the more than electronegative of the ii. Table 1 shows these bonds in order of increasing polarity.
| Bond | ΔEN | Polarity |
|---|---|---|
| C–H | 0.four | [latex]\overset{\delta -}{\text{C}} - \overset{\delta +}{\text{H}}[/latex] |
| S–H | 0.4 | [latex]\overset{\delta -}{\text{South}} - \overset{\delta +}{\text{H}}[/latex] |
| C–North | 0.v | [latex]\overset{\delta +}{\text{C}} - \overset{\delta -}{\text{Northward}}[/latex] |
| N–H | 0.9 | [latex]\overset{\delta -}{\text{N}} - \overset{\delta +}{\text{H}}[/latex] |
| C–O | 1.0 | [latex]\overset{\delta +}{\text{C}} - \overset{\delta -}{\text{O}}[/latex] |
| O–H | 1.4 | [latex]\overset{\delta -}{\text{O}} - \overset{\delta +}{\text{H}}[/latex] |
| Table i. Bond Polarity and Electronegativity Divergence | ||
Check Your Learning
Silicones are polymeric compounds containing, among others, the following types of covalent bonds: Si–O, Si–C, C–H, and C–C. Using the electronegativity values in Figure three, arrange the bonds in guild of increasing polarity and designate the positive and negative atoms using the symbols δ+ and δ–.
Answer:
| Bond | Electronegativity Difference | Polarity |
|---|---|---|
| C–C | 0.0 | nonpolar |
| C–H | 0.4 | [latex]\overset{\delta -}{\text{C}} - \overset{\delta +}{\text{H}}[/latex] |
| Si–C | 0.seven | [latex]\overset{\delta +}{\text{Si}} - \overset{\delta -}{\text{C}}[/latex] |
| Si–O | 1.7 | [latex]\overset{\delta +}{\text{Si}} - \overset{\delta -}{\text{O}}[/latex] |
| Table 2. | ||
Cardinal Concepts and Summary
Covalent bonds class when electrons are shared betwixt atoms and are attracted by the nuclei of both atoms. In pure covalent bonds, the electrons are shared equally. In polar covalent bonds, the electrons are shared unequally, as 1 cantlet exerts a stronger force of attraction on the electrons than the other. The power of an atom to attract a pair of electrons in a chemic bond is chosen its electronegativity. The difference in electronegativity between two atoms determines how polar a bail will be. In a diatomic molecule with ii identical atoms, in that location is no difference in electronegativity, then the bond is nonpolar or pure covalent. When the electronegativity departure is very big, every bit is the instance between metals and nonmetals, the bonding is characterized as ionic.
Chemistry End of Chapter Exercises
- Why is it incorrect to speak of a molecule of solid NaCl?
- What information tin you lot use to predict whether a bond between two atoms is covalent or ionic?
- Predict which of the post-obit compounds are ionic and which are covalent, based on the location of their elective atoms in the periodic tabular array:
(a) Cl2CO
(b) MnO
(c) NCl3
(d) CoBrtwo
(e) G2S
(f) CO
(g) CaF2
(h) Hullo
(i) CaO
(j) IBr
(k) CO2
- Explain the deviation betwixt a nonpolar covalent bond, a polar covalent bond, and an ionic bond.
- From its position in the periodic tabular array, decide which atom in each pair is more electronegative:
(a) Br or Cl
(b) North or O
(c) S or O
(d) P or S
(e) Si or North
(f) Ba or P
(g) N or K
- From its position in the periodic table, make up one's mind which atom in each pair is more electronegative:
(a) N or P
(b) N or Ge
(c) Due south or F
(d) Cl or S
(eastward) H or C
(f) Se or P
(g) C or Si
- From their positions in the periodic table, accommodate the atoms in each of the following series in guild of increasing electronegativity:
(a) C, F, H, N, O
(b) Br, Cl, F, H, I
(c) F, H, O, P, Due south
(d) Al, H, Na, O, P
(eastward) Ba, H, N, O, As
- From their positions in the periodic table, adapt the atoms in each of the following series in order of increasing electronegativity:
(a) Equally, H, N, P, Sb
(b) Cl, H, P, S, Si
(c) Br, Cl, Ge, H, Sr
(d) Ca, H, K, N, Si
(eastward) Cl, Cs, Ge, H, Sr
- Which atoms can bond to sulfur so as to produce a positive fractional accuse on the sulfur atom?
- Which is the virtually polar bond?
(a) C–C
(b) C–H
(c) N–H
(d) O–H
(e) Se–H
- Identify the more than polar bond in each of the following pairs of bonds:
(a) HF or HCl
(b) NO or CO
(c) SH or OH
(d) PCl or SCl
(e) CH or NH
(f) Then or PO
(g) CN or NN
- Which of the following molecules or ions contain polar bonds?
(a) O3
(b) Seight
(c) O22−O22−
(d) NO3−NO3−
(e) CO2
(f) HtwoS
(1000) BH4−BH4−
Glossary
- bond length
- distance between the nuclei of two bonded atoms at which the everyman potential free energy is achieved
- covalent bond
- bond formed when electrons are shared between atoms
- electronegativity
- tendency of an cantlet to attract electrons in a bond to itself
- polar covalent bond
- covalent bond betwixt atoms of different electronegativities; a covalent bond with a positive end and a negative end
- pure covalent bond
- (also, nonpolar covalent bond) covalent bail between atoms of identical electronegativities
Solutions
Answers to Chemical science End of Chapter Exercises
one. NaCl consists of detached ions arranged in a crystal lattice, not covalently bonded molecules.
3. ionic: (b), (d), (eastward), (g), and (i); covalent: (a), (c), (f), (h), (j), and (k)
5. (a) Cl; (b) O; (c) O; (d) S; (e) N; (f) P; (g) N
vii. (a) H, C, North, O, F; (b) H, I, Br, Cl, F; (c) H, P, Due south, O, F; (d) Na, Al, H, P, O; (e) Ba, H, Every bit, N, O
9. N, O, F, and Cl
eleven. (a) HF; (b) CO; (c) OH; (d) PCl; (east) NH; (f) PO; (g) CN
highsmithapereens.blogspot.com
Source: https://opentextbc.ca/chemistry/chapter/7-2-covalent-bonding/
0 Response to "The Number of Electrons in the Valence Shell Determine What Types of Bonds an Atom Can Form"
Post a Comment